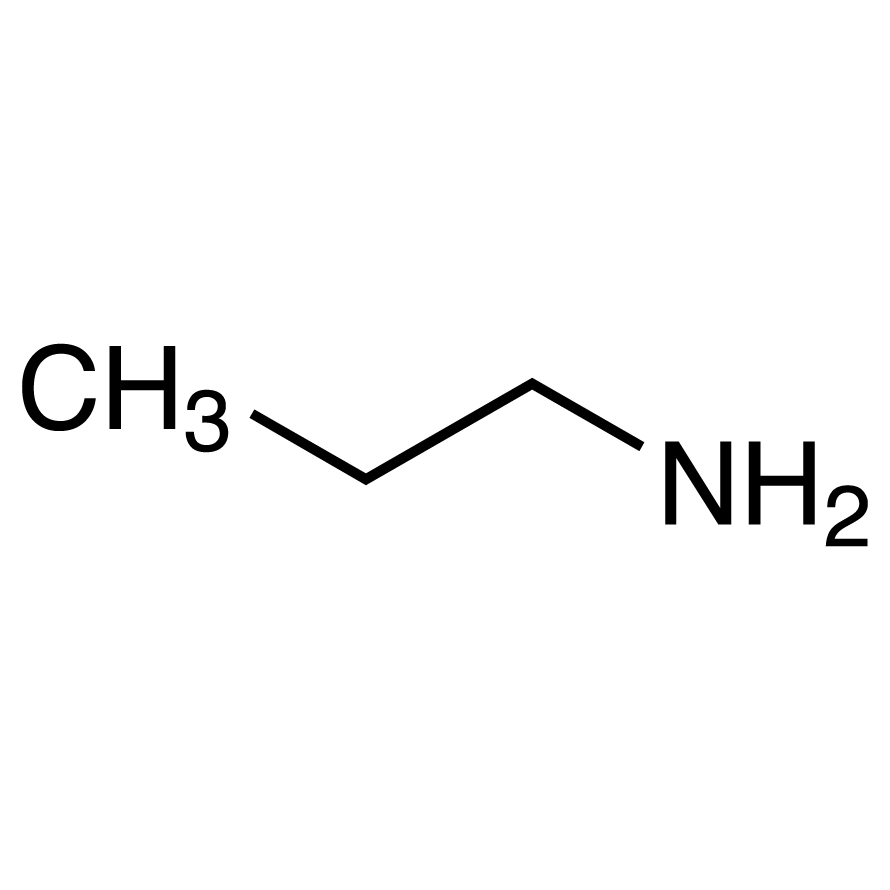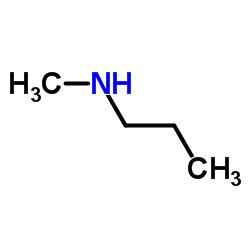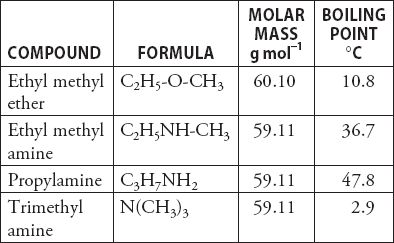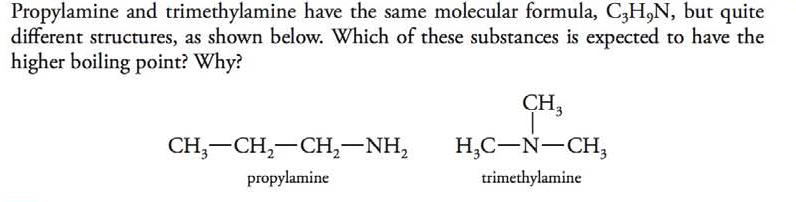
Propylamine and trimethylamine have the same molecular formula, C 3 H 9 N, but quite different structures, as shown below. Which of these substances is expected to have the higher boiling point?
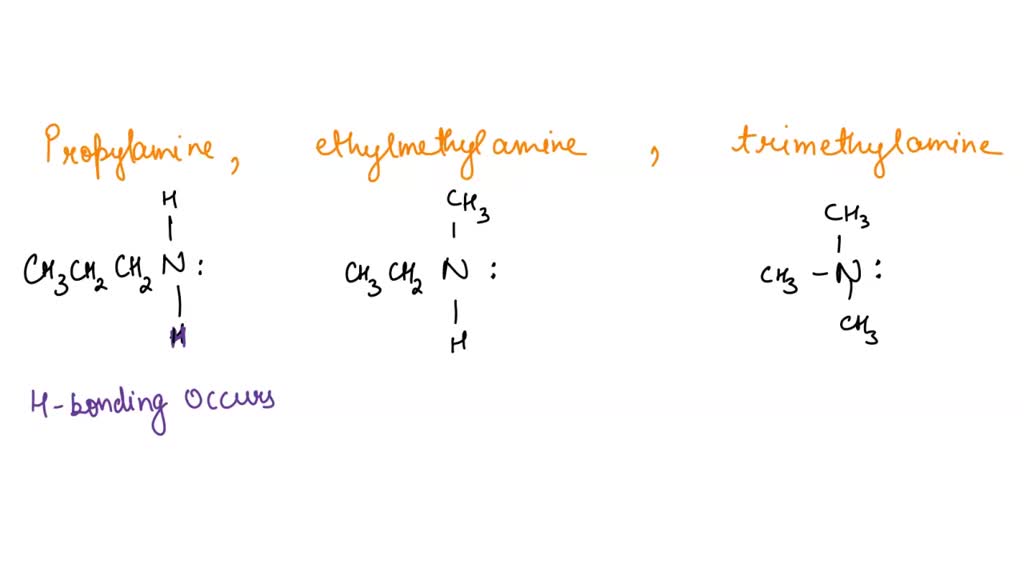
SOLVED: Q131. Propylamine, ethylmethylamine, and trimethylamine have the same molecular formula, CzHgN Explain why the boiling point of trimethylamine is considerably lower than the boiling points of the other two compounds

Linear aliphatic primary amines melting points boiling points solubility in water hydrogen bonding structure classification physical properties of aliphatic amines organic nitrogen compounds advanced A level organic chemistry revision notes doc brown

SOLVED: Q131. Propylamine, ethylmethylamine, and trimethylamine have the same molecular formula, CzHgN Explain why the boiling point of trimethylamine is considerably lower than the boiling points of the other two compounds

Among the following amines namely ethylmethyl amine, propyl amine trimetyl amine, the lowest boi... - YouTube
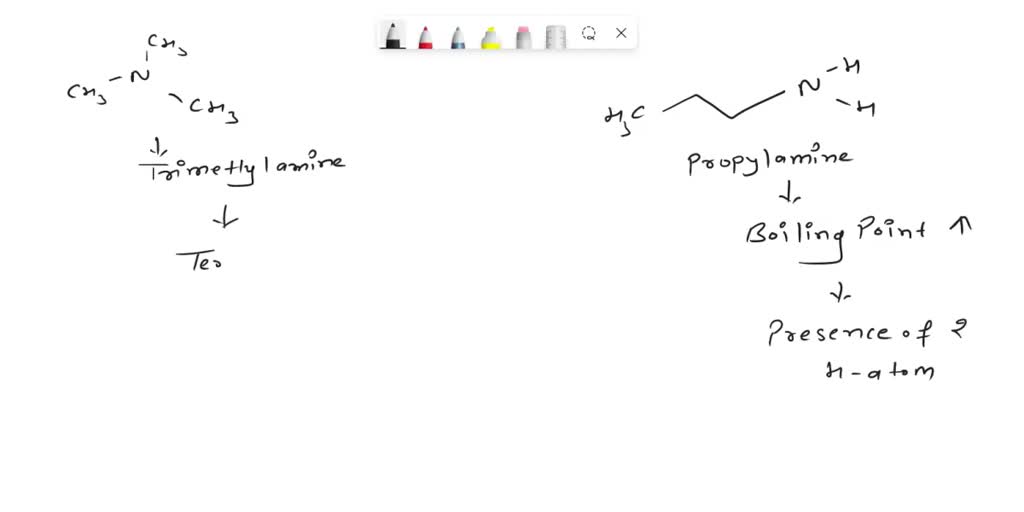

-propylamine[818550_(Trimethoxysilyl)-propylamine-ALL].jpg)