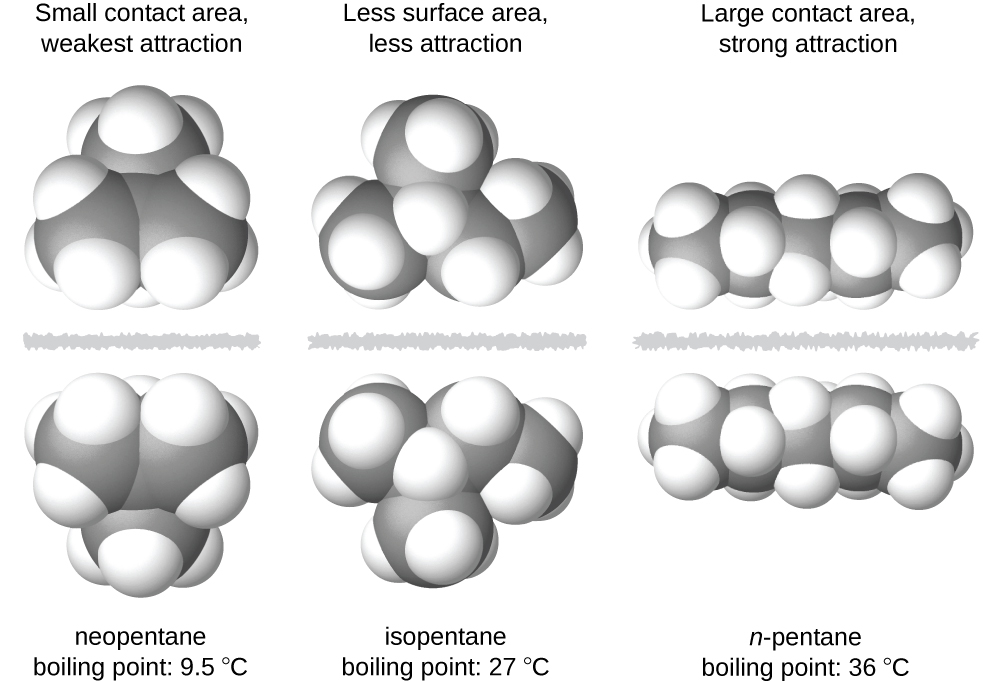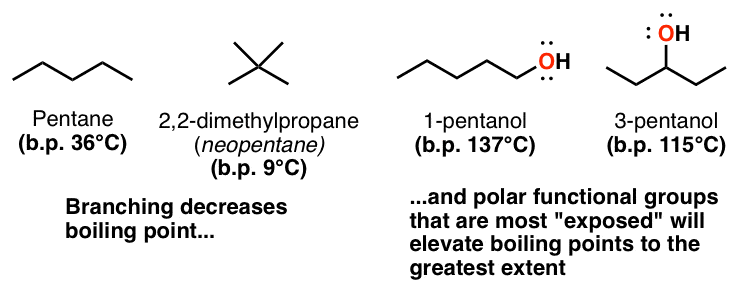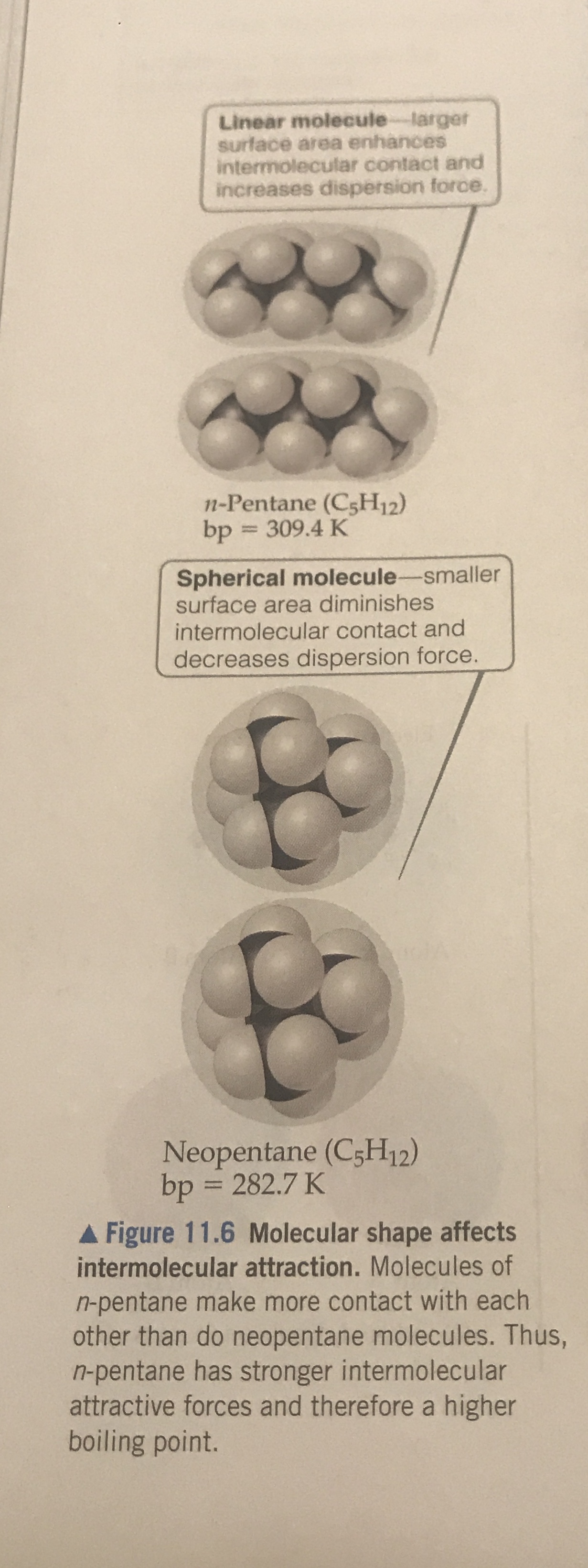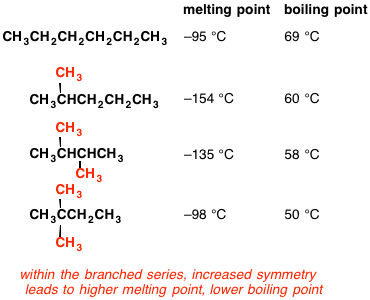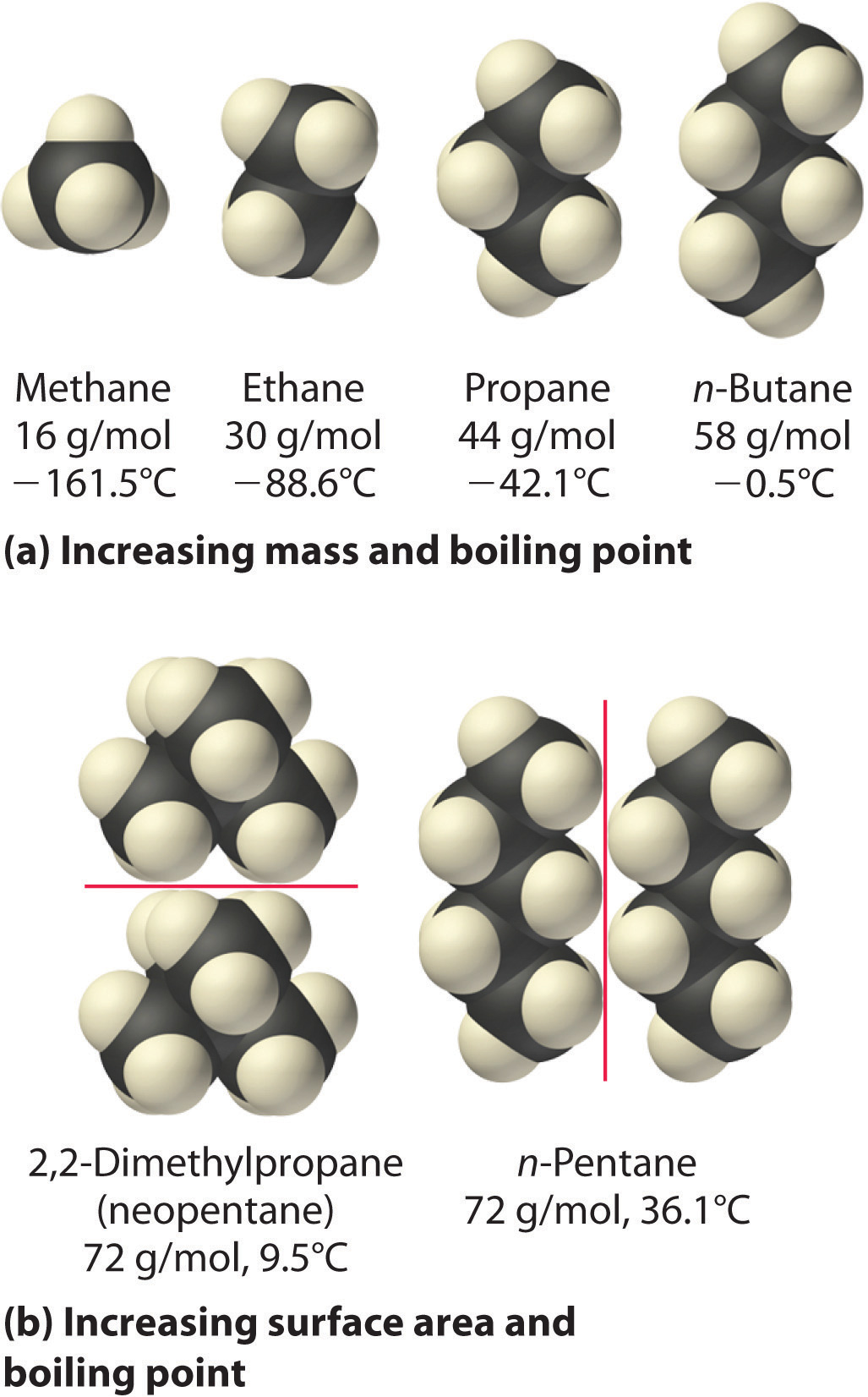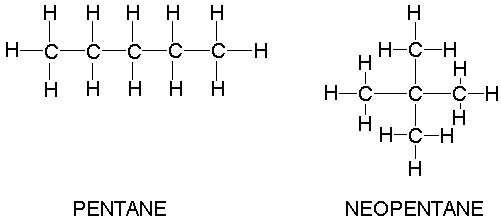
Why when the shape of molecules become more compact it's boiling point decrease while when intermolecular force become strong boiling point increase? | Socratic

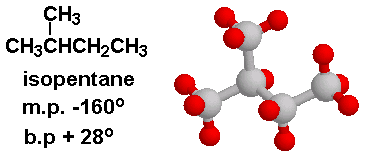
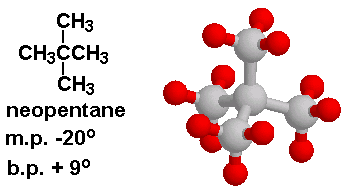
Arrange the following compounds in the descending order of their boiling pointsa) n - pentaneb) isopentanec) neopentane
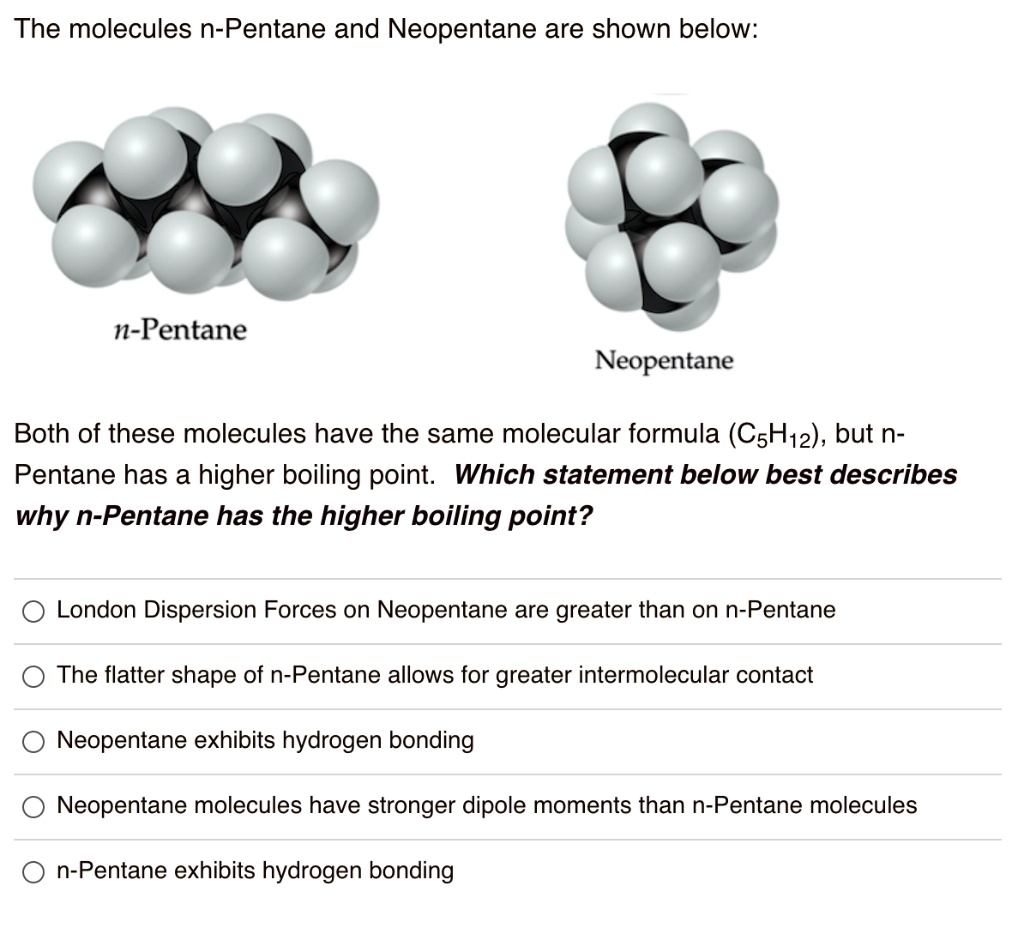
SOLVED: The molecules n-Pentane and Neopentane are shown below: n-Pentane Neopentane Both of these molecules have the same molecular formula (C5H12), but n- Pentane has a higher boiling point: Which statement below
21. Why The boiling point of pentane is greater than isopentane? And why the boiling point of neopentane is less than N pentane and isopentane?
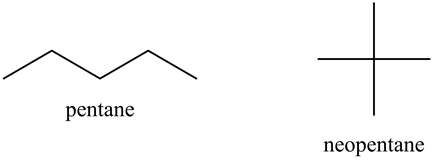
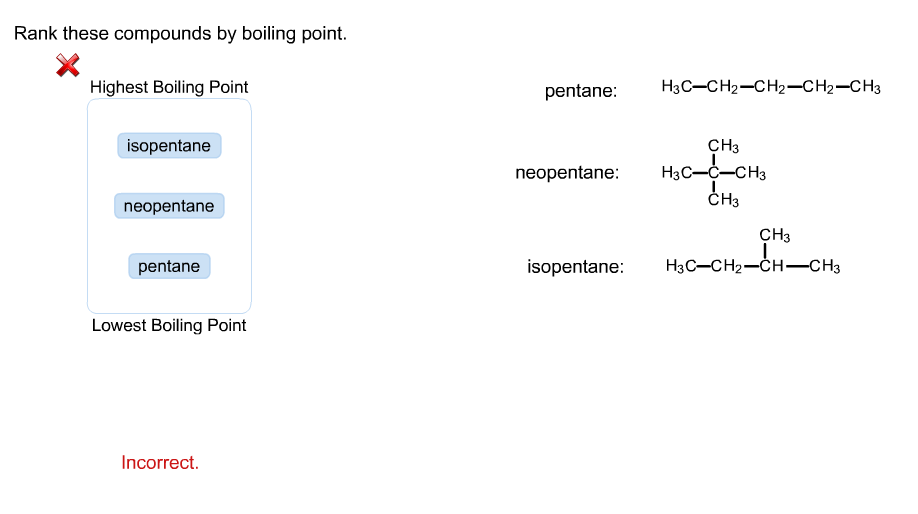
Rank these compounds by boiling point from highest to lowest boiling point: pentane, neopentane, hexane - Home Work Help - Learn CBSE Forum

Arrange the following compounds in the descending order of their boiling pointsa) n - pentaneb) isopentanec) neopentane

SOLVED: Rank the shown compounds by boiling point, pcntanc: HjCCH2=CH=CHz-CH Highest boiling point CH; neopentane: Hsc C CH CH3 CH; isopentane: HsC CHz-Ch CH3 Lowest boiling - point Ansiver Bank pentane isopentane
Why Isomers of a compound have different Boiling point (like Isomers of pentane) why force of attraction is not involve in it? - Quora