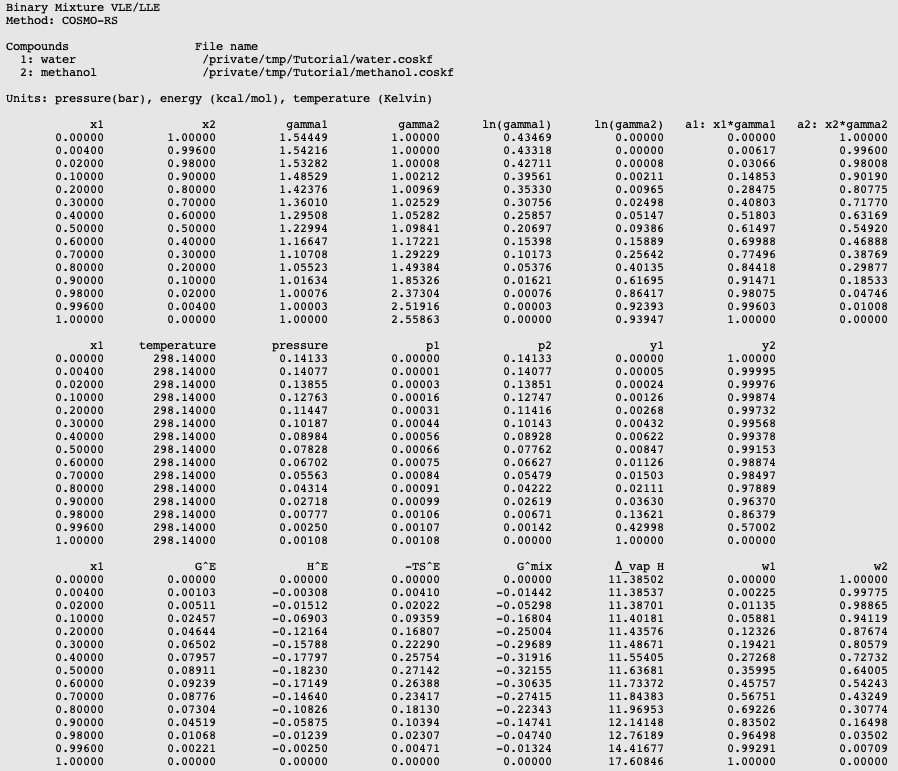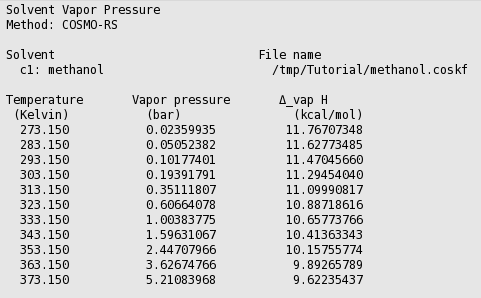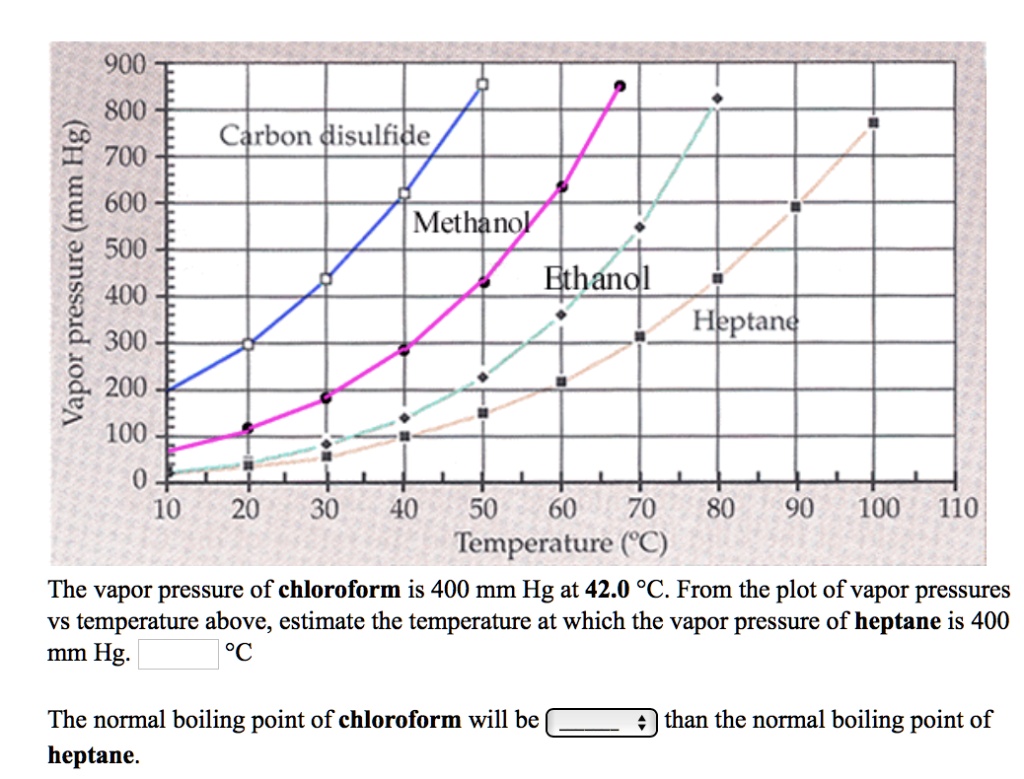
SOLVED: 900 8Q0 2 Carbon disulfide 700 1 600 Methanol 500 L Ethanol 400 Heptand 300 200 100 0 10 20 30 40 50 60 70 80 90 100 110 Temperature (C)

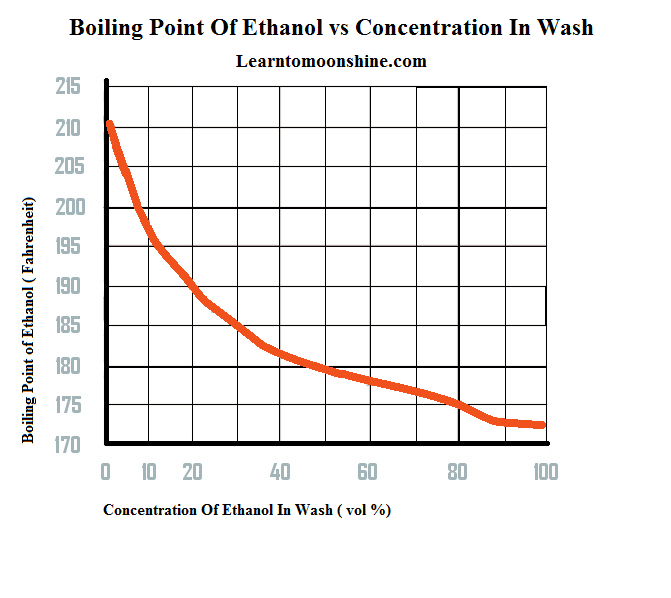
See: Sketch a heating curve graph of temperature The b.p. of methanol is 65°C and that of ethanol is 78°C. - Brainly.com

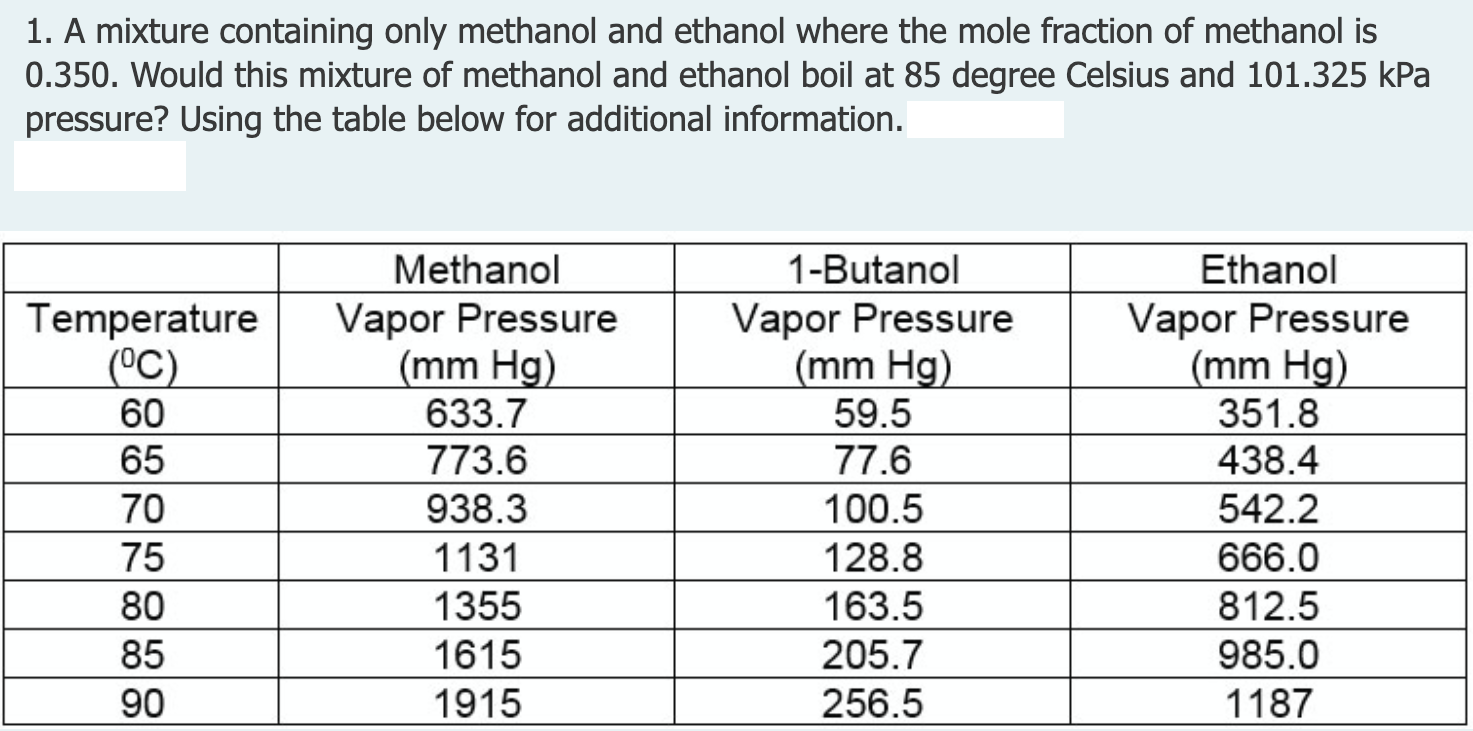
A benzene-methanol system shows azeotropic behavior. After solving for the boiling points for both molecules, describe the behavior for a mixture that is initially rich in benzene (90%) and then for a

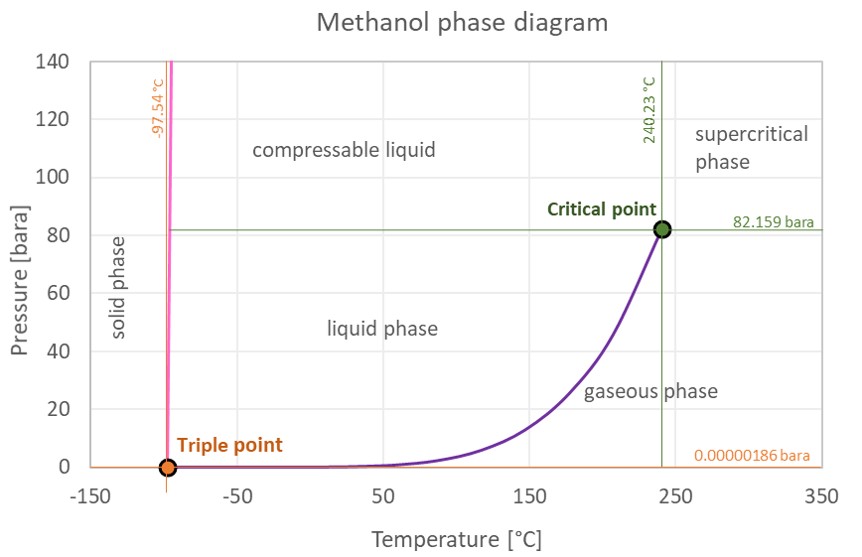
a) Phase diagram of methanol shows route 1 employed in a conventional... | Download Scientific Diagram

SOLVED:The boiling point of methanol is 65.0^∘ C and the standard enthalpy of formation of methanol vapor is -201.2 kJ / mol . Calculate the vapor pressure of methanol (in mmHg )

Whiskey Destilliert Stockfoto und mehr Bilder von Destillation - Destillation, Whisky, Labor - iStock

SOLVED: Methanol (CH3OH) has an enthalpy of vaporization of 35.2 kJ/mol and a normal boiling point of 64.7 degrees C. What is the vapor pressure (in torr) of methanol at 30 degrees

Methanol vapor pressure curve. Markers located at atmospheric pressure... | Download Scientific Diagram