Name the following(1) Thte non - sublimable solid from a mxiture of iodine and potassion nitrate.(2) The heavier liquid component from - mercury and water(3) The lower boiling point component from methyl

The boiling point of butan-2-one (80 C) is significantly higher than the boiling point of diethyl ether (35 C), even though both compounds exhibit dipole-dipole interactions and have comparable molecular weights. Offer

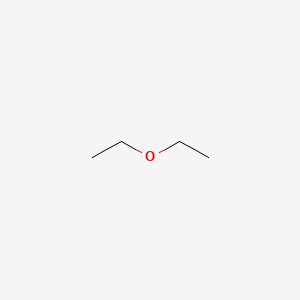
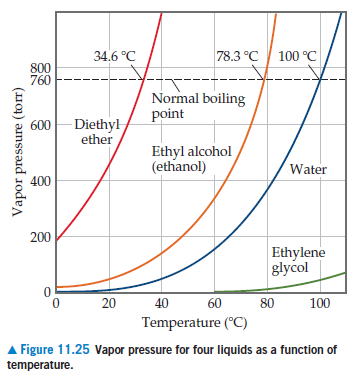
The boiling point of water, ethyl alcohol and diethyl ether are `100^(@)C, 78.5^(@)C` respectively. - YouTube

Diethyl ether has a much higher boiling point than butane despite having a higher molecular weight. Explain why this is the case, making reference to the molecular structures of both compounds.

Diethyl ether has a normal boiling point of `35.0^(@)C` and has an entropy of vaporization of `84.4 - YouTube

Diethyl ether - An SDS for a substance is not primarily intended for use by the general consumer, - Studocu