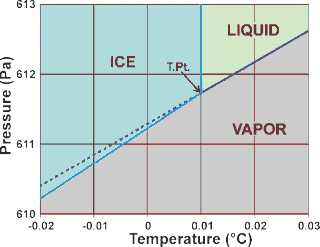
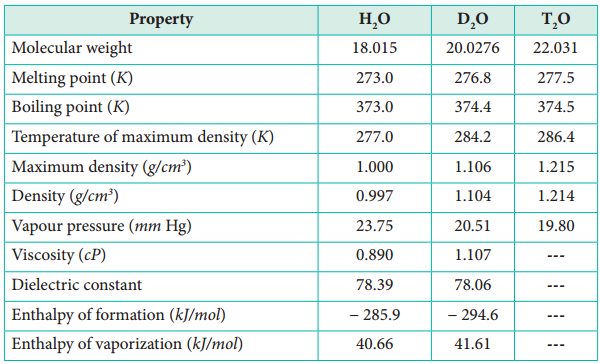
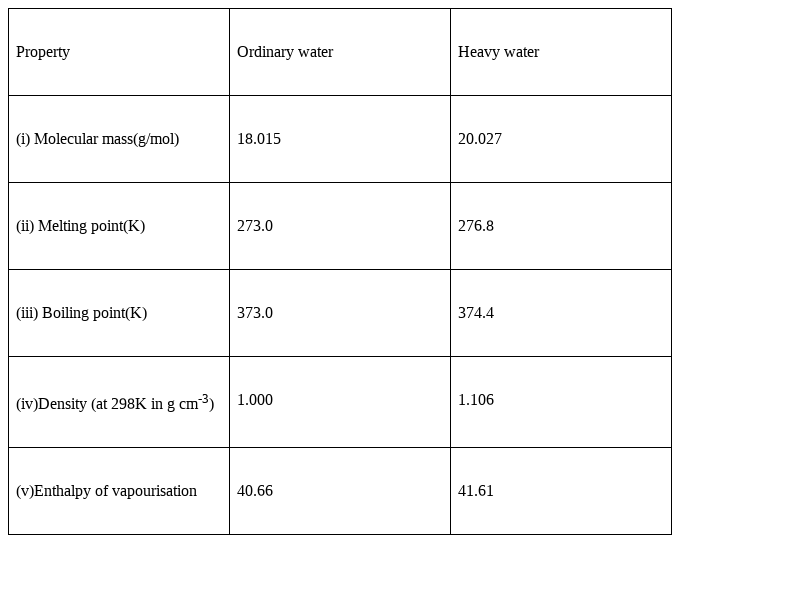
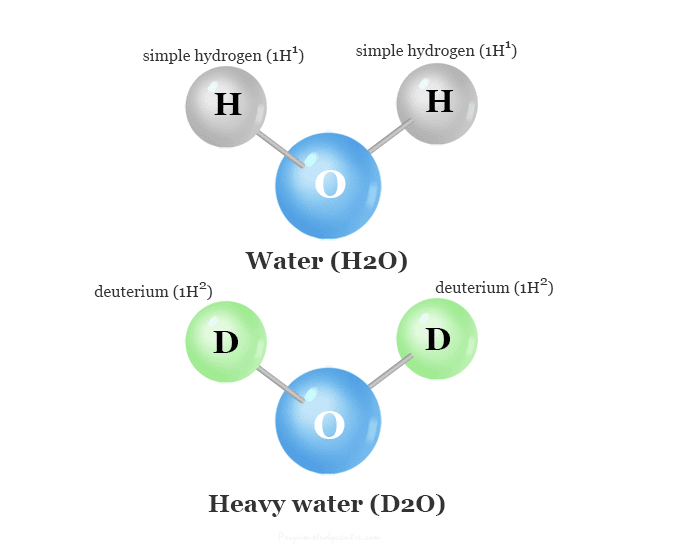
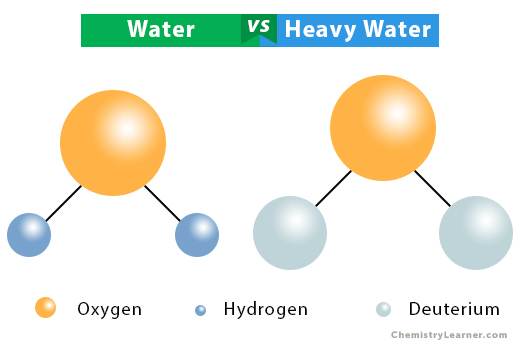
Some statements about heavy water are given below.(i) Heavy water is used as moderator in nuclear reactors(ii) Heavy water is more associated then ordinary water(iii) Heavy water is more effective solvent than
How can D2O be prepared from water? Mention the physical properties in which D2O differs from H2O. - Sarthaks eConnect | Largest Online Education Community